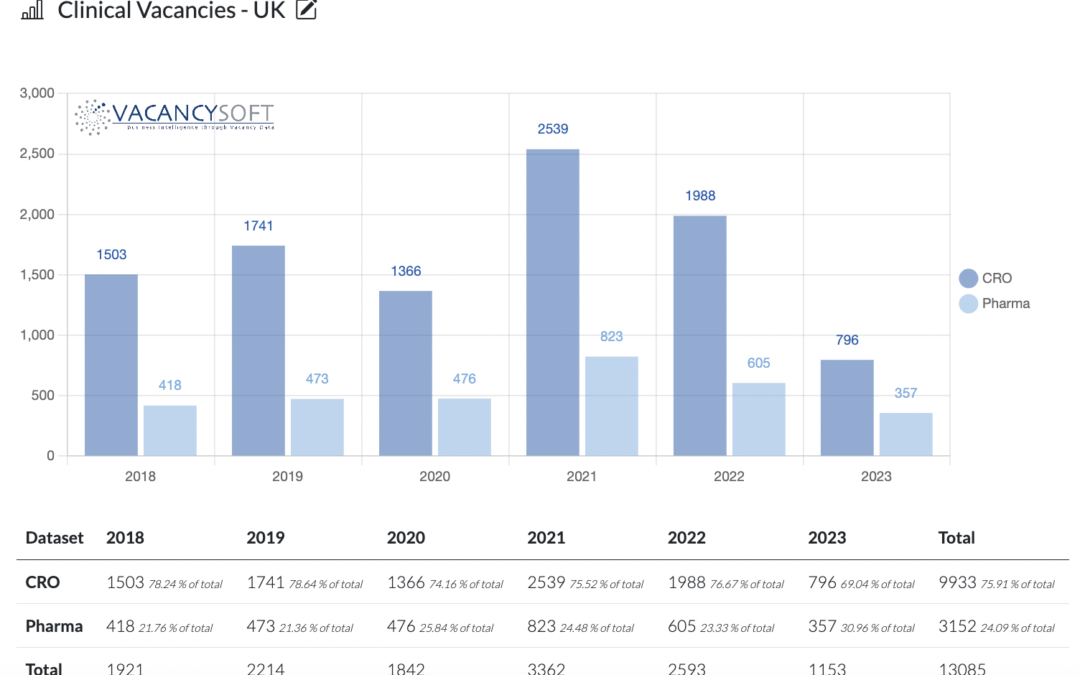
The UK Life Sciences labour market is undergoing major shifts, with AI adoption, regulatory reforms, and regional realignment driving demand for new skills. London leads the resurgence, while nationwide hiring trends reflect growing confidence in the MHRA’s post-Brexit innovation agenda.

The 2023 year saw a dip across both DACH and BENELUX regions, reflected by the EU data. DACH has kept higher numbers than BENELUX since the start of 2022, but this gap is now shortening whilst still being apparent. So far, the first half 2024 is projected to see lower volumes than the full-year estimate.

As the UK moves past the pandemic, the life sciences industry is still adjusting to the challenges associated with the post-Brexit regime. This, combined with the slowdown in trials during the pandemic, has resulted in a slowdown in recruitment within the sector, particularly affecting regulatory affairs professionals.
Whisper it quietly, but some of those benefits of leaving the EU are actually starting to materialize. For those in Life Sciences, new regulations taking effect have meant that the MHRA can now fast-track applications, where as a result, approvals can happen in certain instances, within 14 days.
Vacancies for regulatory affairs professionals across UK life sciences fell in the first months of 2022, following a year when pharmaceutical firms published the most new regulatory jobs since records began, new research shows.